Chinese Journal of Tissue Engineering Research ›› 2023, Vol. 27 ›› Issue (17): 2772-2779.doi: 10.12307/2023.067
Previous Articles Next Articles
Platelet-rich plasma meets the therapeutic needs of different articular cartilage defects
Zhang Ziyu1, 2, Wang Yiming2, Li Han2, Wang Zhonghan3, Lu Jialin2, Xu Rui2, 4, Jin Hui1
- 1Department of Pain Treatment, 3Department of Orthopedics, The Second Hospital of Jilin University, Changchun 130041, Jilin Province, China; 2Norman Bethune Health Science Center of Jilin University, Changchun 130021, Jilin Province, China; 4Department of Endocrinology and Metabolism, Ruijin Hospital, Shanghai Jiao Tong University School of Medicine, Shanghai 200025, China
-
Received:2022-01-05Accepted:2022-04-18Online:2023-06-18Published:2022-10-25 -
Contact:Jin Hui, MD, Attending physician, Department of Pain Treatment, The Second Hospital of Jilin University, Changchun 130041, Jilin Province, China -
About author:Zhang Ziyu, Department of Pain Treatment, The Second Hospital of Jilin University, Changchun 130041, Jilin Province, China; Norman Bethune Health Science Center of Jilin University, Changchun 130021, Jilin Province, China -
Supported by:The Interdisciplinary Research Funding Program for Doctoral Candidates of Jilin University, No. 419100200861 (to WZH)
CLC Number:
Cite this article
Zhang Ziyu, Wang Yiming, Li Han, Wang Zhonghan, Lu Jialin, Xu Rui, Jin Hui. Platelet-rich plasma meets the therapeutic needs of different articular cartilage defects[J]. Chinese Journal of Tissue Engineering Research, 2023, 27(17): 2772-2779.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks
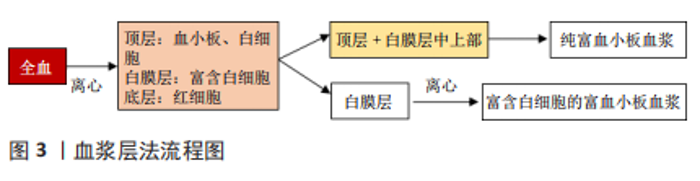
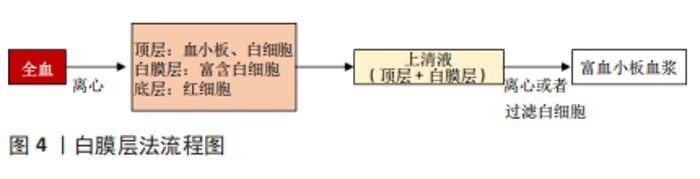
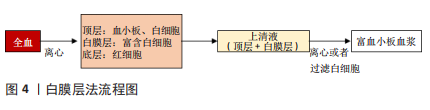
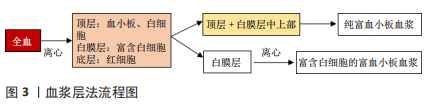
2.1 富血小板血浆的使用 2.1.1 富血小板血浆的制备 目前研究中常用的制备方法有2种,分别为血浆层法和白膜层法,见图3,4。血浆层法中,全血第1次离心分离红细胞,第2次离心浓缩血小板。第1次离心后,试管内液体分为3层,顶层为贫血小板和白细胞,中间层富含白细胞和血小板的薄层称为白膜层,底层主要由红细胞组成。针对所需的富血小板血浆的要求,在第1次离心后进行不同的提取步骤:若需纯富血小板血浆则提取上层和白膜层中上部的液体;若需富含白细胞的富血小板血浆则提取整个白膜层。随后将被提取的液体进行高速离心,最终所获液体下部1/3为富血小板血浆。白膜层法中,第1次高速离心全血后,液体形成了3层,取出上清血浆即顶层和白膜层后低速离心或使用白细胞过滤器分离白细胞即可得到富血小板血浆[17]。"
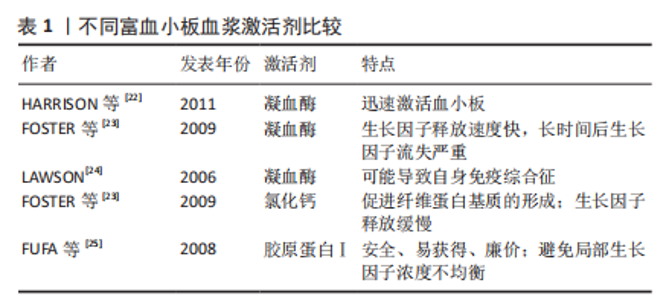
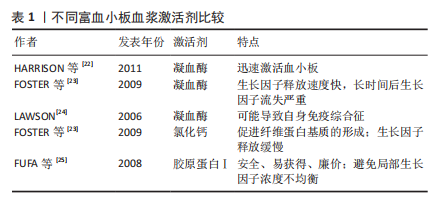
2.1.2 富血小板血浆的储存 富血小板血浆通常在制备后立即投入使用。然而,在需少量多次富血小板血浆注射的治疗方案中,富血小板血浆制备后的储存可以避免多次提取全血造成的疼痛和经济负担[18]。富血小板血浆的短时储存通常在-30 ℃进行,使用前在37 ℃的干燥恒温器中解冻30 min即可投入使用,这种方法可以充分保持诱导软骨细胞增殖和分泌细胞外基质的能力[19]。长时储存通常在-80 ℃进行,有研究表明采用这种储存方法在1个月内可以尽可能多地保持血小板衍生生长因子和转化生长因子β1浓度不变,但胰岛素样生长因子1浓度会降低,如果胰岛素样生长因子1是治疗中必要的生长因子,制备后储存富血小板血浆则是弊大于利[20]。 2.1.3 富血小板血浆的激活 激活剂促进了富血小板血浆中的血小板脱颗粒并释放生长因子的过程 [21]。目前普遍使用的激活剂有3种:凝血酶、氯化钙和胶原蛋白Ⅰ,见表1。对于半衰期短的生长因子来说,若从血小板中释放出后不立即使用,则会很快发生降解,凝血酶能迅速激活血小板[22]。但经凝血酶激活的富血小板血浆在局部生长因子的总量随着时间的推移明显减少,无法长时间保证受损组织所需的生长因子浓度[23]。另外,凝血酶会促进抗凝血酶、凝血酶原、因子V和心磷脂的抗体产生,这可能会进一步导致严重的不良反应,如严重术后出血、旁路移植血栓和类似狼疮的自身免疫综合征等[24]。氯化钙能够使血小板内生长因子缓慢释放且对人体无害,故可作为凝血酶的替代物激活富血小板血浆。氯化钙可促进纤维蛋白基质的形成,纤维蛋白基质具有捕获血小板的特性,故可使血小板最小程度地活化,缓慢释放内含的生长因子,利于生长因子浓度的维持。另外,纤维蛋白基质本身可作为支架促进细胞迁移和细胞外基质的形成从而促进受损组织的愈合[23]。胶原蛋白Ⅰ安全、易获得且廉价,也是凝血酶的替代物之一。与凝血酶相比,胶原蛋白Ⅰ能够防止富血小板血浆凝块过度收缩,避免了受损组织局部生长因子浓度的不均衡,利于关节软骨缺损的修复[25]。"
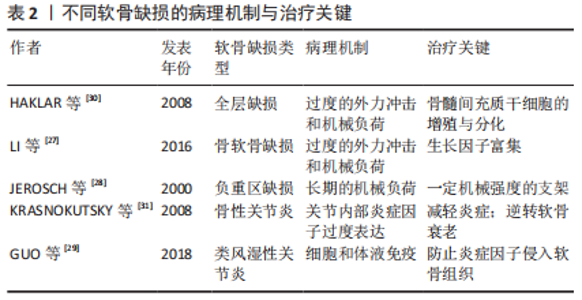
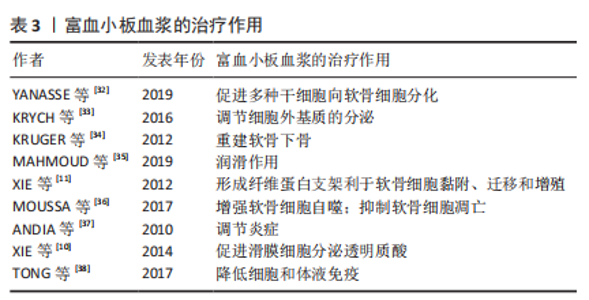
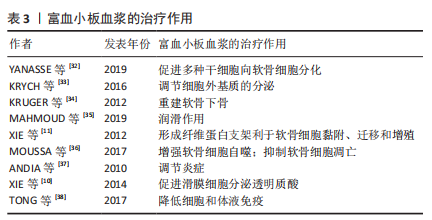
2.2 富血小板血浆治疗软骨缺损 根据软骨缺损的深度可将关节软骨缺损分为全层软骨缺损和骨软骨缺损,根据缺损位置的负重情况可分为负重区缺损和非负重区缺损[26]。全层软骨缺损会对软骨甚至软骨下组织造成损伤,若软骨下骨出现缺损则称为骨软骨缺损。不同的病理机制导致不同的软骨缺损。过度的外力冲击和机械负荷可能导致全层关节软骨缺损,甚至骨软骨缺损[27]。承受长期机械负荷的负重区软骨更易受损[28]。由于肥胖、性别、衰老等多种因素造成的关节内部炎症因子过度表达则会刺激软骨降解进而导致关节炎的进展[29]。促进软骨细胞的增殖、诱导干细胞分化为软骨细胞以及调节软骨细胞的分泌是治疗以上软骨缺损的共同需求[30-31],见表2,除此之外,不同的软骨缺损还有不同的治疗需求。结合富血小板血浆治疗软骨缺损的研究,针对不同软骨缺损的病理机制阐述富血小板血浆对缺损软骨的修复作用,见表3[11-12,32-38]。 2.2.1 深度缺损 (1)全层缺损:是深及关节软骨全层的损伤,由于有血液渗入缺损处,全层缺损的软骨具有一定的自我修复能力。 血液中的骨髓间充质干细胞具有向软骨细胞分化的潜能,在受到损伤后将形成类似透明软骨样组织,但这种组织以Ⅰ型胶原为主,而健康关节软骨组织以Ⅱ型胶原为主[39]。这种自愈形成的组织与健康软骨组织不完全相同,无法完全恢复关节软骨的生物学和力学性能,最终仍会导致关节功能异常。故全层软骨损伤的修复不能仅依靠自愈,必须使再生软骨的成分尽可能接近健康软骨才可恢复关节功能。多项研究表明富血小板血浆能够提高再生软骨的质量从而促进全层软骨缺损的修复,与自愈形成的组织相比更接近透明软骨[32,40-41]。然而,也有部分研究对富血小板血浆修复全层软骨缺损能力的评价较为负面,他们认为富血小板血浆不能显著促进透明软骨的再生[42]。尽管如此,富血小板血浆应用于临床治疗全层软骨缺损仍具有很大的可能性。HEDE等[43]的临床研究表明富血小板血浆治疗全层软骨损伤可改善术后1,2年的主观临床疗效评分和疼痛评分。还有临床试验表明富血小板血浆可以促进损伤处的软骨再生并且再生软骨的成分与形态接近透明软骨[33]。这些临床研究肯定了富血小板血浆促进全层软骨的再生能力。除此之外,富血小板血浆能促进多种干细胞向软骨细胞分化[32]。骨髓间充质干细胞作为一种具有分化潜能的干细胞,不仅能在富血小板血浆的存在下显著提高增殖与分化能力,还可通过旁分泌释放多种利于软骨修复的生长因子,包括肝细胞生长因子、胰岛素样生长因子1、转化生长因子β、碱性成纤维细胞生长因子等[44]。 故富血小板血浆对全层软骨缺损的修复作用满足其治疗需求,主要体现在2个方面:一是富血小板血浆对骨髓间充质干细胞增殖与分化的促进作用;二是富血小板血浆对软骨细胞分泌细胞外基质的调节作用,这改善了再生软骨组织的成分,弥补了自愈产生的再生软骨中胶原蛋白Ⅰ含量过高的缺陷,提高了再生软骨的质量,利于关节功能恢复。 (2)骨软骨缺损:全层软骨缺损若进一步加深至软骨下骨则会导致骨软骨缺损,包括骨、软骨和骨-软骨界面的损伤或变性[45]。骨软骨缺损周围的软骨细胞外基质中糖胺聚糖含量减少,纤维软骨形成增加,导致关节功能受损[46]。与其他类型的缺损不同,骨软骨缺损的修复需要重建带状关节软骨和软骨下骨,恢复与天然骨软骨组织一致的结构和生物力学性能[47]。目前普遍使用自体移植术,但不能完全恢复受损组织的结构与功能[48]。有研究证明,转化生长因子、胰岛素样生长因子、骨形态发生蛋白等生长因子在骨软骨再生中发挥重要作用,这些生长因子可改善软骨层细胞的排列和蛋白多糖的沉积,并且诱导软骨下骨层的新骨形成[49]。因此,骨软骨缺损的治疗关键在于缺损处多种生长因子的富集。富血小板血浆作为富含多种生长因子的自体生物制剂,能够影响骨软骨的代谢过程,对骨软骨缺损的治疗具有很强的针对性。多项研究验证了富血小板血浆的治疗能力:对于软骨层,富血小板血浆促进软骨细胞分泌糖胺聚糖和胶原蛋白Ⅱ,并且使再生组织结构完整、细胞和胶原排列良好、细胞形态与正常软骨细胞相似,即再生软骨的质量较高 [41,50-51];对于软骨下骨,富血小板血浆能够增强软骨下骨的间充质祖细胞的迁移能力并且刺激其分泌蛋白多糖和Ⅱ型胶原,促进成骨细胞分化、血管生成和骨髓细胞募集,即富血小板血浆具有重建软骨下骨的潜力[34]。整体来看,富血小板血浆具有治疗骨软骨缺损的能力[52-54]。 富血小板血浆在以上两方面的修复作用弥补了当前疗法不能完全恢复受损组织结构的不足,更好地恢复了受损组织的功能,然而,骨软骨缺损修复需要的生长因子的种类和浓度需要进一步明确,富血小板血浆疗法也应据此作出进一步优化。 2.2.2 负重区缺损 根据关节面的受力情况,可将关节面分为负重区和非负重区。人体用于主要负重的关节有3个,从上至下依次为髋关节、膝关节、踝关节:膝关节的内侧和外侧髁属于负重区,而髁间窝和髁上窝属于非负重区;髋关节负重区为髋臼和股骨头;踝关节负重区为距骨和胫骨远端。负重区的关节软骨传递和吸收更多的压力,故比非负重区更易受损[28]。与非负重区相比,负重区的关节软骨更厚,软骨组织中的蛋白多糖含量更高,胶原纤维的排列更为均匀,软骨细胞也更大[55]。由于负重区的特殊受压状态,其缺损的治疗需要特定结构和机械性能的支架[56]。 除了对软骨细胞增殖的促进作用和对再生细胞外基质质量的提高作用,富血小板血浆对负重区软骨缺损的修复作用主要体现在2个方面,一是富血小板血浆的润滑作用。粗糙的关节面会持续损伤软骨组织,缺损软骨修复的过程中需要润滑物质减少关节面的进一步磨损[35,57]。分泌表面区蛋白是关节软骨中的一种边界润滑剂,在减少摩擦和磨损方面起着重要作用,是维持关节软骨内稳态的关键因素。富血小板血浆不仅含有内源性表面区蛋白,而且对关节软骨细胞分泌表面区蛋白有显著促进作用[58]。由于负重区关节软骨更易被磨损,富血小板血浆的润滑作用可减轻负重区关节软骨的磨损以抑制缺损进展。二是富血小板血浆可参与形成具有合适机械性能的支架,适于治疗处于特殊受压状态的负重区软骨。富血小板血浆本身含有纤维蛋白原和其他凝血因子,可被激活形成临时纤维蛋白支架,供软骨细胞黏附、迁移和增殖[11]。LIU等[40]在富血小板血浆中加入光响应透明质酸获得了原位光交联富血小板血浆水凝胶,经实验验证,这种富血小板血浆水凝胶是一种坚固的水凝胶支架,它不仅可以控制生长因子的释放,还表现出很强的组织黏附能力,非常利于负重区软骨的再生。整体来看,富血小板血浆治疗负重区软骨缺损具有很大潜力。多项研究证实了富血小板血浆治疗负重区关节软骨缺损的有效性[50,54,59-61]。 过度的机械负荷会导致关节微环境缺氧,因此负重区缺损的软骨通常都会缺氧,这会导致软骨细胞活力降低甚至凋亡。脂肪源性间充质干细胞获能不依赖有氧呼吸,对缺氧环境耐受力强,并且在缺氧环境下仍具有较强的分化潜能,这使其在修复软骨缺损的种子细胞中具有很大优势[62]。因此,当负重区软骨缺损情况较为严重时,采用富血小板血浆与脂肪源性间充质干细胞联合治疗可能会有较好疗效。适当的机械刺激可以转化为生化信号来调节细胞行为,从而促进软骨再生,但过度负重可能导致促炎细胞因子增加、细胞外基质分泌减少和软骨细胞凋亡[63]。故富血小板血浆治疗后患者的最适负荷训练还需进一步探索。 2.2.3 伴随炎症的软骨缺损 炎症环境下的软骨缺损不同于机械缺损,炎症导致多种细胞因子平衡失调和免疫紊乱,显著改变关节的微环境。若不抑制其进展则会导致关节软骨退化、软骨下骨缺损、关节间隙狭窄和骨质疏松等严重后果[64]。 (1)骨性关节炎:是一种退行性关节疾病,可逐渐导致关节功能丧失甚至身体残疾[31]。目前主要采用物理疗法和关节置换术治疗骨性关节炎,这些疗法可延缓软骨降解的进展但不能修复受损的软骨[65]。关节超负荷会破坏关节内环境稳态导致炎症,是骨性关节炎的主要诱因[66]。炎症因子导致细胞内各种细胞因子平衡失调、诱导软骨细胞凋亡、减少软骨细胞外基质沉积、影响胶原纤维排列,进一步破坏骨性关节炎软骨[67]。年龄与骨性关节炎密切相关,随着年龄增长,摩擦损伤逐渐累积、软骨细胞合成分泌活性降低、细胞表面信号受体减少等导致细胞对生长因子的敏感度降低、软骨组织再生能力降低,最终内稳态失衡,导致关节内环境出现炎症[68]。故除促进软骨再生外,治疗骨性关节炎的关键在于逆转软骨衰老和减轻炎症。 富血小板血浆可促进骨性关节炎软骨细胞的自噬,逆转软骨细胞衰老自噬和凋亡相互关联,富血小板血浆影响细胞自噬同时引起了软骨细胞凋亡基因的改变,显著降低了B细胞淋巴瘤2相关死亡启动子(Bad)和半胱天冬酶3(Caspase-3)的mRNA水平并提高B细胞淋巴瘤2(Bcl-2)的mRNA水平,抑制了软骨细胞的凋亡[36]。故富血小板血浆具有抑制软骨衰老的作用,利于骨性关节炎软骨缺损的治疗。同时,富血小板血浆具有调节炎症的作用,其可通过抑制白细胞介素1β介导的多种炎症因子的表达减轻关节内环境的炎症反应[69]。除此之外,富血小板血浆中的富生长因子血小板能显著促进滑膜细胞分泌透明质酸[70]。透明质酸作为滑液中的重要成分能够润滑和营养关节,并且可减轻骨性关节炎疼痛[71]。简而言之,富血小板血浆的作用满足了骨性关节炎的治疗需求,在修复骨性关节炎软骨缺损方面具有很大潜力。 (2)类风湿关节炎:是最常见的慢性全身炎症性疾病,属于自身免疫性疾病,约占全球人口的1%,进一步发展可能导致软骨退化、骨缺损、残疾甚至过早死亡[72]。目前常用疗法为药物治疗和手术治疗,这些疗法只能延缓软骨降解的进展而不能修复受损的软骨[73],并且存在缺陷:药物治疗可能会引起感染、间质性肺病和糖代谢紊乱等不良反应[74];手术治疗创伤大、术后粘连广泛,并且不可重复进行[75]。类风湿关节炎确切的致病机制仍不明确,就目前所知,细胞和体液免疫参与类风湿关节炎的发生与进展,主要致病因子是来自M1巨噬细胞的白细胞介素1、白细胞介素6和肿瘤坏死因子α,这些因子通过刺激软骨细胞和成纤维细胞释放基质金属蛋白酶分解软骨细胞外基质,并通过激活破骨细胞引起骨吸收,最终导致软骨损伤[29]。因此其治疗主要依赖于微环境的免疫调节,关键在于防止炎症因子侵入软骨组织[76]。巨噬细胞主要分为M1和M2表型:M1巨噬细胞介导宿主对各种细菌、原生动物和病毒的防御,具有促进炎症反应的作用;M2巨噬细胞具有抗炎和调节伤口愈合的作用。M1巨噬细胞向M2巨噬细胞的转变是类风湿关节炎软骨修复的关键[77]。 转化生长因子β1能促进M1巨噬细胞向M2巨噬细胞转化,并在M2巨噬细胞的活化中起关键作用[78]。富血小板血浆中含有较高浓度的转化生长因子β1,经实验证明,富血小板血浆具有促进M1巨噬细胞转化为M2巨噬细胞的能力[79]。另外,即使在类风湿关节炎条件下,富血小板血浆仍可通过释放血小板衍生生长因子BB、白细胞介素6和肿瘤坏死因子等蛋白维持软骨细胞外基质的胶原含量以减轻软骨损伤[9]。由此可得,富血小板血浆可防止炎症因子侵入软骨组织,符合类风湿关节炎软骨缺损的治疗需求,具有治疗潜力。值得注意的是,巨噬细胞的表型转化会受富血小板血浆所含白细胞浓度的影响,贫白细胞富血小板血浆(LP-PRP)较富白细胞富血小板血浆(LR-PRP)更多地诱导M1巨噬细胞转化为M2巨噬细胞[80]。因此在富血小板血浆的制备过程中应注意控制白细胞浓度。除此之外,富血小板血浆还具有降低体液和细胞免疫反应的能力[38]。多项体内实验证明富血小板血浆治疗类风湿关节炎是有效的[38,81]。这些实验研究验证了富血小板血浆治疗类风湿关节炎软骨缺损的能力,但由于类风湿关节炎的致病机制尚不明确,富血小板血浆治疗类风湿关节炎的机制也无法完全确定。 "
| [1] CARBALLO CB, NAKAGAWA Y, SEKIYA I, et al. Basic Science of Articular Cartilage. Clin Sport Med. 2017;36(3):413-425. [2] TESHIMA R, NAWATA K, HAGINO H, et al. Effects of weight bearing on the tidemark and osteochondral junction of articular cartilage: histomorphometric analyses of 7 normal femoral heads. Acta Orthop Scand. 1999;70(4):381-386. [3] LAKSTINS K, ARNOLD L, GUNSCH G, et al. Characterization of bovine and canine animal model cartilage endplates and comparison to human cartilage endplate structure, matrix composition, and cell phenotype. JOR Spine. 2020;3(4):e1116. [4] SILVERBERG JL, BARRETT AR, DAS M, et al. Structure-function relations and rigidity percolation in the shear properties of articular cartilage. Biophys J. 2014;107(7):1721-1730. [5] LE H, XU W, ZHUANG X, et al. Mesenchymal stem cells for cartilage regeneration. J Tissue Eng. 2020;11:2041731420943839. [6] O’DRISCOLL SW. The healing and regeneration of articular cartilage. J Bone Joint Surg Am. 1998;80(12):1795-1812. [7] LAMPLOT JD, SCHAFER KA, MATAVA MJ. Treatment of Failed Articular Cartilage Reconstructive Procedures of the Knee: A Systematic Review. Orthop J Sports Med. 2018;6(3):2325967118761871. [8] ZHU Y, YUAN M, MENG HY, et al. Basic science and clinical application of platelet-rich plasma for cartilage defects and osteoarthritis: a review. Osteoarthritis Cartilage. 2013;21(11):1627-1637. [9] LIPPROSS S, MOELLER B, HAAS H, et al. Intraarticular injection of platelet-rich plasma reduces inflammation in a pig model of rheumatoid arthritis of the knee joint. Arthritis Rheum. 2011;63(11):3344-3353. [10] XIE X, ZHANG C, TUAN RS. Biology of platelet-rich plasma and its clinical application in cartilage repair. Arthritis Res Ther. 2014;16(1):204. [11] XIE X, WANG Y, ZHAO C, et al. Comparative evaluation of MSCs from bone marrow and adipose tissue seeded in PRP-derived scaffold for cartilage regeneration. Biomaterials. 2012;33(29):7008-7018. [12] BORN GV. Evidence for the formation of a labile phospholipoprotein during the clotting of platelet-rich plasma. Nature. 1957;180(4585): 546-547. [13] EVERTS PA, KNAPE JT, WEIBRICH G, et al. Platelet-rich plasma and platelet gel: a review. J Extra Corpor Technol. 2006;38(2):174-187. [14] MCCARREL T, FORTIER L. Temporal growth factor release from platelet-rich plasma, trehalose lyophilized platelets, and bone marrow aspirate and their effect on tendon and ligament gene expression. J Orthop Res. 2009;27(8):1033-1042. [15] SAKATA R, REDDI AH. Platelet-Rich Plasma Modulates Actions on Articular Cartilage Lubrication and Regeneration. Tissue Eng Part B Rev. 2016;22(5):408-419. [16] GIUFFRE G, CAPUTO G, MISSO S, et al. Platelet-rich plasma treatment and hemostasis in patients with hemorrhagic risk. Minerva Stomatol. 2006;55(11-12):599-609. [17] DHURAT R, SUKESH M. Principles and Methods of Preparation of Platelet-Rich Plasma: A Review and Author’s Perspective. J Cutan Aesthet Surg. 2014;7(4):189-197. [18] SMETS F, CROISIER JL, FORTHOMME B, et al. Clinical applications of platelet-rich plasma (PRP) in tendon lesions: A literature review. Sci Sport. 2012;27(3):141-153. [19] ROFFI A, FILARDO G, ASSIRELLI E, et al. Does platelet-rich plasma freeze-thawing influence growth factor release and their effects on chondrocytes and synoviocytes. Biomed Res Int. 2014;2014:692913. [20] MCCLAIN AK, MCCARREL TM. The effect of four different freezing conditions and time in frozen storage on the concentration of commonly measured growth factors and enzymes in equine platelet-rich plasma over six months. Bmc Vet Res. 2019;15(1):292. [21] BRICKSON S, JI LL, SCHELL K, et al. M1/70 attenuates blood-borne neutrophil oxidants, activation, and myofiber damage following stretch injury. J Appl Physiol (1985). 2003;95(3):969-976. [22] HARRISON S, VAVKEN P, KEVY S, et al. Platelet activation by collagen provides sustained release of anabolic cytokines. Am J Sports Med. 2011;39(4):729-734. [23] FOSTER TE, PUSKAS BL, MANDELBAUM BR, et al. Platelet-rich plasma: from basic science to clinical applications. Am J Sports Med. 2009;37(11):2259-2272. [24] LAWSON JH. The clinical use and immunologic impact of thrombin in surgery. Semin Thromb Hemost. 2006;32 Suppl 1:98-110. [25] FUFA D, SHEALY B, JACOBSON M, et al. Activation of platelet-rich plasma using soluble type I collagen. J Oral Maxillofac Surg. 2008;66(4):684-690. [26] NUKAVARAPU SP, DORCEMUS DL. Osteochondral tissue engineering: Current strategies and challenges. Biotechnology Advances. 2013; 31(5):706-721. [27] LI X, WEI J, AIFANTIS KE, et al. Current investigations into magnetic nanoparticles for biomedical applications. J Biomed Mater Res A. 2016; 104(5):1285-1296. [28] JEROSCH J, FILLER T, PEUKER E. Is there an option for harvesting autologous osteochondral grafts without damaging weight-bearing areas in the knee joint. Knee Surg Sports Traumatol Arthrosc. 2000;8(4): 237-240. [29] GUO Q, WANG Y, XU D, et al. Rheumatoid arthritis: pathological mechanisms and modern pharmacologic therapies. Bone Res. 2018;6:15. [30] HAKLAR U, TUZUNER T, UYGUR I, et al. The effect of overlapping on the primary stability of osteochondral grafts in mosaicplasty. Knee Surg Sports Traumatol Arthrosc. 2008;16(7):651-654. [31] KRASNOKUTSKY S, ATTUR M, PALMER G, et al. Current concepts in the pathogenesis of osteoarthritis. Osteoarthritis Cartilage. 2008;16 Suppl 3:S1-S3. [32] YANASSE RH, DE LABIO RW, MARQUES L, et al. Xenotransplantation of human dental pulp stem cells in platelet-rich plasma for the treatment of full-thickness articular cartilage defects in a rabbit model. Exp Ther Med. 2019;17(6):4344-4356. [33] KRYCH AJ, NAWABI DH, FARSHAD-AMACKER NA, et al. Bone Marrow Concentrate Improves Early Cartilage Phase Maturation of a Scaffold Plug in the Knee: A Comparative Magnetic Resonance Imaging Analysis to Platelet-Rich Plasma and Control. Am J Sport Med. 2016;44(1):91-98. [34] KRUGER JP, HONDKE S, ENDRES M, et al. Human platelet-rich plasma stimulates migration and chondrogenic differentiation of human subchondral progenitor cells. J Orthop Res. 2012;30(6):845-852. [35] MAHMOUD EE, ADACHI N, MAWAS AS, et al. Coculturing of mesenchymal stem cells of different sources improved regenerative capability of osteochondral defect in the mature rabbit: An in vivo study. J Orthop Surg (Hong Kong). 2019;27(2):2309499019839850. [36] MOUSSA M, LAJEUNESSE D, HILAL G, et al. Platelet rich plasma (PRP) induces chondroprotection via increasing autophagy, anti-inflammatory markers, and decreasing apoptosis in human osteoarthritic cartilage. Exp Cell Res. 2017;352(1):146-156. [37] ANDIA I, SANCHEZ M, MAFFULLI N. Tendon healing and platelet-rich plasma therapies. Expert Opin Biol Ther. 2010;10(10):1415-1426. [38] TONG S, ZHANG C, LIU J. Platelet-rich plasma exhibits beneficial effects for rheumatoid arthritis mice by suppressing inflammatory factors. Mol Med Rep. 2017;16(4):4082-4088. [39] MARTIN JA, BUCKWALTER JA. The role of chondrocyte-matrix interactions in maintaining and repairing articular cartilage. Biorheology. 2000;37(1-2):129-140. [40] LIU XL, YANG YL, NIU X, et al. An in situ photocrosslinkable platelet rich plasma - Complexed hydrogel glue with growth factor controlled release ability to promote cartilage defect repair. Acta Biomater. 2017;62: 179-187. [41] CHANG NJ, ERDENEKHUYAG Y, CHOU PH, et al. Therapeutic Effects of the Addition of Platelet-Rich Plasma to Bioimplants and Early Rehabilitation Exercise on Articular Cartilage Repair. Am J Sports Med. 2018;46(9):2232-2241. [42] OLESEN ML, CHRISTENSEN BB, FOLDAGER CB, et al. No Effect of Platelet-Rich Plasma Injections as an Adjuvant to Autologous Cartilage Chips Implantation for the Treatment of Chondral Defects. Cartilage. 2021;13(2_suppl):277S-284S. [43] HEDE K, CHRISTENSEN BB, JENSEN J, et al. Combined Bone Marrow Aspirate and Platelet-Rich Plasma for Cartilage Repair: Two-Year Clinical Results. Cartilage. 2021;13(1_suppl):937S-947S. [44] TOGEL F, WEISS K, YANG Y, et al. Vasculotropic, paracrine actions of infused mesenchymal stem cells are important to the recovery from acute kidney injury. Am J Physiol Renal Physiol. 2007;292(5): F1626-F1635. [45] LOPA S, MADRY H. Bioinspired Scaffolds for Osteochondral Regeneration. Tissue Eng Pt A. 2014;20(15-16):2052-2076. [46] LIN X, CHEN J, QIU P, et al. Biphasic hierarchical extracellular matrix scaffold for osteochondral defect regeneration. Osteoarthritis Cartilage. 2018;26(3):433-444. [47] ZHOU F, CHU L, LIU X, et al. Subchondral Trabecular Microstructure and Articular Cartilage Damage Variations Between Osteoarthritis and Osteoporotic Osteoarthritis: A Cross-sectional Cohort Study. Front Med (Lausanne). 2021;8:617200. [48] ORTH P, REY-RICO A, VENKATESAN JK, et al. Current perspectives in stem cell research for knee cartilage repair. Stem Cells Cloning. 2014; 7:1-17. [49] KIM K, LAM J, LU S, et al. Osteochondral tissue regeneration using a bilayered composite hydrogel with modulating dual growth factor release kinetics in a rabbit model. J Control Release. 2013;168(2):166-178. [50] SMYTH NA, HALEEM AM, ROSS KA, et al. Platelet-Rich Plasma May Improve Osteochondral Donor Site Healing in a Rabbit Model. Cartilage. 2016;7(1):104-111. [51] ZHANG YT, NIU J, WANG Z, et al. Repair of Osteochondral Defects in a Rabbit Model Using Bilayer Poly(Lactide-co-Glycolide) Scaffolds Loaded with Autologous Platelet-Rich Plasma. Med Sci Monitor. 2017;23: 5189-5201. [52] SCIOLI MG, BIELLI A, GENTILE P, et al. Combined treatment with platelet-rich plasma and insulin favours chondrogenic and osteogenic differentiation of human adipose-derived stem cells in three-dimensional collagen scaffolds. J Tissue Eng Regen Med. 2017;11(8): 2398-2410. [53] AIRAPETOV GA, VOROTNIKOV AA, VENEDIKTOV AA, et al. Replacement of Osteochondral Defects of Major Joints in Experiment. Sovrem Tehnol Med. 2019;11(3):55-58. [54] YAN W, XU X, XU Q, et al. Platelet-rich plasma combined with injectable hyaluronic acid hydrogel for porcine cartilage regeneration: a 6-month follow-up. Regen Biomater. 2020;7(1):77-90. [55] BAGI CM, ZAKUR DE, BERRYMAN E, et al. Correlation between muCT imaging, histology and functional capacity of the osteoarthritic knee in the rat model of osteoarthritis. J Transl Med. 2015;13:276. [56] VAN DER WOUDE JAD, WIEGANT K, VAN ROERMUND PM, et al. Five-Year Follow-up of Knee Joint Distraction: Clinical Benefit and Cartilaginous Tissue Repair in an Open Uncontrolled Prospective Study. Cartilage. 2017;8(3):263-271. [57] CHANG NJ, LIN YT, LIN CC, et al. The repair of full-thickness articular cartilage defect using intra-articular administration of N-acetyl-D-glucosamine in the rabbit knee: randomized controlled trial. Biomed Eng Online. 2015;14:105. [58] SAKATA R, MCNARY SM, MIYATAKE K, et al. Stimulation of the superficial zone protein and lubrication in the articular cartilage by human platelet-rich plasma. Am J Sports Med. 2015;43(6):1467-1473. [59] BETSCH M, SCHNEPPENDAHL J, THUNS S, et al. Bone marrow aspiration concentrate and platelet rich plasma for osteochondral repair in a porcine osteochondral defect model. Plos One. 2013;8(8):e71602. [60] AKPANCAR S, GUL D. Comparison of Platelet Rich Plasma and Prolotherapy in the Management of Osteochondral Lesions of the Talus: A Retrospective Cohort Study. Med Sci Monit. 2019;25: 5640-5647. [61] GU WQ, LI TZ, SHI ZM, et al. Management of Hepple Stage V Osteochondral Lesion of the Talus with a Platelet-Rich Plasma Scaffold. Biomed Res Int. 2017;2017:6525373. [62] ZHOU W, LIN J, ZHAO K, et al. Single-Cell Profiles and Clinically Useful Properties of Human Mesenchymal Stem Cells of Adipose and Bone Marrow Origin. Am J Sports Med. 2019;47(7):1722-1733. [63] DAVIS S, ROLDO M, BLUNN G, et al. Influence of the Mechanical Environment on the Regeneration of Osteochondral Defects. Front Bioeng Biotechnol. 2021;9:603408. [64] FREITAG J, BATES D, BOYD R, et al. Mesenchymal stem cell therapy in the treatment of osteoarthritis: reparative pathways, safety and efficacy - a review. BMC Musculoskelet Disord. 2016;17:230. [65] NALESSO G, SHERWOOD J, BERTRAND J, et al. WNT-3A modulates articular chondrocyte phenotype by activating both canonical and noncanonical pathways. J Cell Biol. 2011;193(3):551-564. [66] SHARMA L. Osteoarthritis of the Knee. N Engl J Med. 2021;384(1): 51-59. [67] ADLER N, SCHOENIGER A, FUHRMANN H. Effects of transforming growth factor-beta and interleukin-1beta on inflammatory markers of osteoarthritis in cultured canine chondrocytes. Am J Vet Res. 2017; 78(11):1264-1272. [68] BLANEY DAVIDSON EN, SCHARSTUHL A, VITTERS EL, et al. Reduced transforming growth factor-beta signaling in cartilage of old mice: role in impaired repair capacity. Arthritis Res Ther. 2005;7(6):R1338-R1347. [69] VAN BUUL GM, KOEVOET WL, KOPS N, et al. Platelet-rich plasma releasate inhibits inflammatory processes in osteoarthritic chondrocytes. Am J Sports Med. 2011;39(11):2362-2370. [70] ANITUA E, SANCHEZ M, NURDEN AT, et al. Platelet-released growth factors enhance the secretion of hyaluronic acid and induce hepatocyte growth factor production by synovial fibroblasts from arthritic patients. Rheumatology (Oxford). 2007;46(12):1769-1772. [71] JANG JD, MOON YS, KIM YS, et al. Novel repair technique for articular cartilage defect using a fibrin and hyaluronic acid mixture. Tissue Eng Regen Med. 2013;10(1):1-9. [72] MCINNES IB, SCHETT G. Pathogenetic insights from the treatment of rheumatoid arthritis. Lancet. 2017;389(10086):2328-2337. [73] DULIC S, VASARHELYI Z, SAVA F, et al. T-Cell Subsets in Rheumatoid Arthritis Patients on Long-Term Anti-TNF or IL-6 Receptor Blocker Therapy. Mediators Inflamm. 2017;2017:6894374. [74] PEIRÓ CADAHÍA J, BONDEBJERG J, HANSEN CA, et al. Synthesis and Evaluation of Hydrogen Peroxide Sensitive Prodrugs of Methotrexate and Aminopterin for the Treatment of Rheumatoid Arthritis. J Med Chem. 2018;61(8):3503-3515. [75] KADOTA Y, NISHIDA K, HASHIZUME K, et al. Risk factors for surgical site infection and delayed wound healing after orthopedic surgery in rheumatoid arthritis patients. Mod Rheumatol. 2016;26(1):68-74. [76] CAO W, CAO K, CAO J, et al. Mesenchymal stem cells and adaptive immune responses. Immunol Lett. 2015;168(2):147-153. [77] ARNOLD L, HENRY A, PORON F, et al. Inflammatory monocytes recruited after skeletal muscle injury switch into antiinflammatory macrophages to support myogenesis. J Exp Med. 2007;204(5):1057-1069. [78] NOVAK ML, KOH TJ. Phenotypic transitions of macrophages orchestrate tissue repair. Am J Pathol. 2013;183(5):1352-1363. [79] LI H, HICKS JJ, WANG L, et al. Customized platelet-rich plasma with transforming growth factor beta1 neutralization antibody to reduce fibrosis in skeletal muscle. Biomaterials. 2016;87:147-156. [80] NISHIO H, SAITA Y, KOBAYASHI Y, et al. Platelet-rich plasma promotes recruitment of macrophages in the process of tendon healing. Regen Ther. 2020;14:262-270. [81] PAN W, DAI C, LI Y, et al. PRP-chitosan thermoresponsive hydrogel combined with black phosphorus nanosheets as injectable biomaterial for biotherapy and phototherapy treatment of rheumatoid arthritis. Biomaterials. 2020;239:119851. [82] DELONG JM, RUSSELL RP, MAZZOCCA AD. Platelet-rich plasma: the PAW classification system. Arthroscopy. 2012;28(7):998-1009. [83] HARRISON P, SUBCOMMITTEE ON PLATELET PHYSIOLOGY. The use of platelets in regenerative medicine and proposal for a new classification system: guidance from the SSC of the ISTH. J Thromb Haemost. 2018; 16(9):1895-1900. [84] LE ADK, ENWEZE L, DEBAUN MR, et al. Current Clinical Recommendations for Use of Platelet-Rich Plasma. Curr Rev Musculoskelet Med. 2018;11(4):624-634. [85] SU CY, KUO YP, NIEH HL, et al. Quantitative assessment of the kinetics of growth factors release from platelet gel. Transfusion. 2008;48(11): 2414-2420. [86] MCDEVITT CA, MUIR H. Biochemical changes in the cartilage of the knee in experimental and natural osteoarthritis in the dog. J Bone Joint Surg Br. 1976;58(1):94-101. [87] SU JW, LI SF, TAO JJ, et al. Estrogen protects against acidosis-mediated articular chondrocyte injury by promoting ASIC1a protein degradation. Eur J Pharmacol. 2021;908:174381. [88] MITRAGOTRI S, YOO JW. Designing micro- and nano-particles for treating rheumatoid arthritis. Arch Pharm Res. 2011;34(11):1887-1897. [89] KRONENBERG HM. Developmental regulation of the growth plate. Nature. 2003;423(6937):332-336. [90] AZARPIRA MR, SHAHCHERAGHI GH, AYATOLLAHI M, et al. Tissue engineering strategy using mesenchymal stem cell-based chitosan scafolds in growth plate surgery: a preliminary study in rabbits. Orthop Traumatol Surg Res. 2015;101(5):601-605. |
| [1] | Li Xiaomin, Tian Xiangdong, Tan Yetong, Zhu Guangyu, Wang Rongtian, Wang Jian, Xue Zhipeng, Ma Sheng, Hu Yuanyi, Huang Ye, Ding Tiansong. Changes of lower limb force line and knee function after high tibial osteotomy in osteoporotic medial ventricular knee osteoarthritis [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(9): 1325-1329. |
| [2] | Zheng Hongrui, Zhang Wenjie, Wang Yunhua, He Bin, Shen Yajun, Fan Lei. Femoral neck system combined with platelet-rich plasma in the treatment of femoral neck fracture [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(9): 1390-1395. |
| [3] | Huang Linke, Wei Linhua, Jiang Jie, Liu Qian, Chen Weiwei. Effects of estrogen combined with treadmill exercise on bone mass and articular cartilage in ovariectomized mice [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(8): 1166-1171. |
| [4] | Lian Shilin, Zhang Yan, Jiang Qiang, Zhang Hanshuo, Li Tusheng, Ding Yu. Interventional effects of whole blood and platelet-rich plasma with different preparation methods on nucleus pulposus cells [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(8): 1199-1204. |
| [5] | Li Long, Li Guangdi, Shi Hao, Deng Keqi. Circular RNA as a competing endogenous RNA is involved in the regulation of osteoarthritis [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(5): 751-757. |
| [6] | Yuan Changshen, Guan Yanbing, Li Zhe, Rong Weiming, Liao Shuning, Chen Lewei, Mei Qijie, Duan Kan. Screening and verification of key genes of necroptosis in osteoarthritis [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(5): 695-700. |
| [7] | Wan Guoli, Shi Chenhui, Wang Weishan, Li Ang, Shi Xunda, Cai Yi. Retrospective analysis of the influencing factors of chronic pain after total knee arthroplasty [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(4): 558-564. |
| [8] | Gu Mingxi, Wang Bo, Tian Fengde, An Ning, Hao Ruihu, Wang Changcheng, Guo Lin. Comparison of early efficacy and safety of simultaneous and staged bilateral total knee arthroplasty [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(4): 565-571. |
| [9] | Yu He, Zheng Jiafa, Song Xiufeng, Guan Shengyi. Tibiotalocalcaneal arthrodesis with blood supplied fibular flap combined with hollow screw in the treatment of end-stage ankle osteoarthritis [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(4): 588-593. |
| [10] | Guo Yingqi, Gong Xianxu, Zhang Yan, Xiao Han, Wang Ye, Gu Wenguang. Meniscus extrusion and patellofemoral joint cartilage injury and bone marrow lesions: MRI semi-quantitative score [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(4): 600-605. |
| [11] | Yu Jiaan, Liu Xinwei, Lian Hongyu, Liu Kexin, Li Zitao. Medial open-wedge tibial osteotomy versus lateral closed-wedge tibial osteotomy for unicompartmental knee osteoarthritis: a meta-analysis [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(4): 632-639. |
| [12] | Ning Ziwen, Wang Xu, Shi Zhengliang, Qin Yihua, Wang Guoliang, Jia Di, Wang Yang, Li Yanlin. Meniscal injury repair methods for non-blood supply area [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(3): 420-426. |
| [13] | Xia Yubo, Tang Xiaoxia, Luo Wen, Xu Yongsheng, Yuan Changfei, Wang Zhe, Zhou Xiaohan, Tian Miao, Wang Tao, Guo Ying. Biomechanical response of ankle traction arthroplasty for ankle osteoarthritis through finite element modeling [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(22): 3469-3475. |
| [14] | Hu Wei, Yan Xianke . Effect of posterior cruciate ligament preserving knee prosthesis on gait and lower extremity venous return in varus knee arthroplasty [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(22): 3503-3507. |
| [15] | Zhong Hehe, Jin Ying, Liu Xiuqi, Xiang Kuan, Wu Shuhong, Peng Jiachen, Liu Yi. High tibial osteotomy combined with arthroscopy to treat degenerative tear in the posterior horn of medial meniscus combined with varus deformity of the knee [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(22): 3531-3536. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||